Project Description
Description of caustic soda flake | NaOH Flake
Caustic soda flakes (sodium hydroxide flakes) are prepared by liquid caustic soda which is produced by membrane cell technology. Therefore, this product has the best quality and is without heavy metal impurities. Caustic soda flakes are highly hygroscopic and soluble in water and are used in a lot of different industries .The purity of this product is min %99. Which is packed with 25kg thick PP/PE bags
FEATURES
Caustic soda flakes use for:
Caustic soda flake , or sodium hydroxide, mostly used by industry and chemical manufacturing companies. Some fields Caustic soda flake use for :
- Refineries Alumina
- plastic wrap
- Soaps and cleaners
- Detergent
- Textile processing
- Oil refining
- Water treatment
- Metal processing
Formula Caustic soda flake:
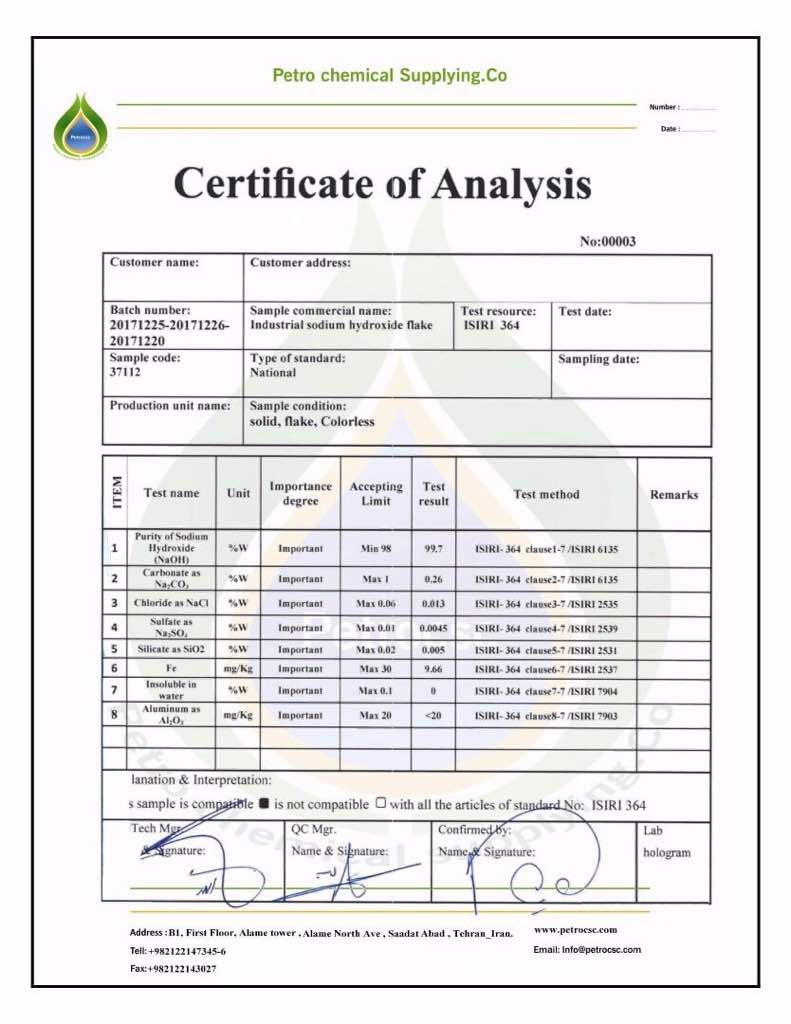
Sodium hydroxide ( NaOH), purity / mass: Min. 99.50
Chlorides / NaCl, purity / mass: Max. 0.10
Sodium carbonate / Na2CO3, purity / mass, Max. 0.40
Caustic Soda Pearls
A caustic soda pearl is a white sphere with a trace odor. Despite being soluble in water by releasing heat, they are stable under normal conditions. Caustic soda pearls are commonly used for:
- In addition to processing cotton, aluminum etching, painting, disrobing, producing soap and detergents, they are used to manufacture petroleum.
- In the paper industry, pearls of caustic soda are used to whiten the paper.
- Aluminum industries use this chemical. The manufacturing of aluminum requires caustic soda as a way to soften bauxite minerals.
- As a result of saponification, Caustic Soda Pearls help to produce vegetable oil and fat for manufacturing soap.
- They can be used in the natural gas industry to produce and process petroleum products and the textile industry for chemically processing cotton.
The Differences Between Caustic Soda Pearls and Caustic Soda Flakes?
The most significant difference between caustic soda pearls and caustic soda flakes is their physical form. The caustic soda pearls are smooth like fish eggs, but the flakes are lamellar in texture that resemble tiny snowflakes.
Soda pearl is less ‘aggressive’ than caustic soda, which contains sodium hydroxide. Because Caustic Soda is regularly used to clear blocked sanitations, a mix-up could occur.
The application of the caustic soda pearls is simple and easy to determine, making them ideal for use in scientific research. On the other hand, a low cost of production makes caustic soda flakes suitable for industrial use.
Different processes are used to produce caustic soda flakes and caustic soda pearls. The chloralkali method involves electrolyzing sodium chloride using either a mercury, diaphragm, or membrane cell to make caustic soda. By contrast, sodium hydroxide flakes are produced by vaporizing liquid caustic soda.
To keep caustic soda pearls from eroding, they should be stored in containers with high corrosion resistance. It is necessary to implement further containment procedures due to the “leak-seeking” nature of NaOH and the potential chemical hazards it poses. Certain metals, including lead, bronze, and aluminum, should not be used when storing or transporting Caustic soda pearls. Be sure that the Caustic Soda Flakes are stored in tightly closed, clearly labeled containers. Since hydrogen gas may be created while caustic soda flakes are stored in aluminum containers, they should not be collected there. Caustic soda flakes should be stored in well-ventilated, dry, cold spaces in order to minimize dust formation and spread. You should keep caustic soda flakes away from acids, ammonium salts, and other ingredients.
Last but not least, it is noteworthy to emphasize that:
Due to its wide range of applications, Caustic soda flake is considered a mother material, and safety tips are required when using this substance.
We have two kind of packing:
First packing is 25kg PP/PE bags in Jumbo Bag (per jumbo bag is weight is 1.250MT). The next packing form is: 25kg PP/PE shrinking on pallet.
Please check album .
In this article, we will answer three essential questions, so stay tuned for the rest of the text.
- caking reasons on caustic soda flakes
- caustic soda flake price
- how to pack caustic soda flake
Caking reasons on caustic soda flake
An inorganic compound called sodium hydroxide, or NaOH, is also known as caustic soda. Sodium cations and hydroxide anion make this white solid ionic compound up. The users noticed caking issues when using sodium hydroxide, but it was unclear what caused these problems. Below are two possible reasons for caustic soda flakes clotting.
The temperature of caustic soda is essential. The flow rate of cooling water and pivoting speed of the machine control the temperature of caustic soda flakes. Caustic soda temperature and consistency of the charging temperature directly depend on the temperature of the caustic soda machine’s water circulation as well as the temperature of the water in the circulation. When the scale arrangement is cleaned after the sprinkler head is cleaned, the circulating water is enlarged, and the temperature of caustic soda flakes is reduced from sixty-five to fifty-eight centigrade degrees, caking building up can be eased.
Laminar bags have a twofold coat and are used as packing bags. Unless the seam is fast enough or if the joint is tight enough, the laminar in the bag will absorb humidity and change its status. The single-seam was transformed into a double seam to accomplish tremendous air isolation and significantly reduce caking problems caused by loose sealing. The packaging staff needed to investigate the entrainment and stitch processes. There is no straight connection between the performance and the caustic soda caking process. Though, if the caking takes place, the standard use and effect of service will be transformed. Hence, the company should pay special attention to users’ products and applications.
caustic soda flakes price
This product (caustic soda flakes) can be purchased in a variety of ways. The market offers a range of prices as well. It is also essential to consider the amount and mode of use in determining the purchase price. There are several elements that influence the cost of caustic soda flakes. For example, a factor that could affect caustic soda flakes’ price is the lack of accessibility to the raw material. In addition, if the dollar price increases in a country, it means that the currency’s value will decrease, and the costs of transport, raw materials, and many other items will rise, impacting the final price of any factory-like caustic soda flakes. Globally, caustic soda flakes products may cost 300 USD or more. Due to its excellent logistics and quality, our company can compete in global markets. The price information is available upon request.
How to pack caustic soda flake
first things first, it is noteworthy to read the safety considerations since the toxic, corrosive properties of Caustic soda flakes make them a hazardous product. Caustic soda flakes lead to severe chemical burns on the skin. Eye and skin irritation is possible. Body contact should be avoided. Taking in dust or mist can seriously harm your respiratory and gastrointestinal systems. A product packaging’s fundamental function is to protect the product it contains. Packages need to ensure product safety during shipping from the manufacturing facility to the retailer and prevent product damage while the product is on the shelf. To that end, packaging should be durable and dependable.
Industrial caustic soda comes in a variety of packaging. HS code 28151100 classifies Caustic Soda Flake as a dangerous good. Caustic soda flakes should be stored in tightly closed containers with appropriate labels. Caustic soda flakes must not be stored in aluminum containers because flammable hydrogen gas can be made. If possible, keep caustic soda flakes in dry and cold, well-ventilated spaces to prevent dust accumulation and spread. Caustic soda flake should never be stored together with other materials, such as acids and salts of ammonium.
Eventually, in the light of this article, we find that:
caustic soda is a white solid ionic compound with a variety of usage. That may ruin by caking issues when you use sodium hydroxide. There are 2 fundamental reasons for this situation, temperature and dampness absorption. First, the flow rate of cooling water and pivoting speed of the machine control the temperature of caustic soda flakes. And for the second, Laminar bags have a twofold coat and are used as packing bags. Unless the seam is fast enough or if the joint is tight enough, the laminar in the bag will simply absorb humidity and change its status. So this implies the important role of packaging. And finally, do not forget to read the safety considerations.
Caustic Soda flakes or Caustic Soda Pearls?
In this article, we discuss the chemistry, administration, and toxicity of Caustic Soda Pearls and Caustic Soda Flakes.
First things first, we should know what caustic soda is. In fact, it is sodium hydroxide (NaOH) by the trading name “caustic soda flakes.” This white, liquid odorless chemical compound is an inorganic substance that belongs to the strongest alkalies, is highly hygroscopic, soluble in water (111 g of soda per 100 ml of water at 20 ºc.), glycerine, methanol, and alcohol. Caustic soda is a foundational raw material in nearly all manufacturing, e.g., paper, soap, exfoliating masks, commercial drain, and oven cleaners.
Caution!!!!
Protective gloves are recommended when working with Caustic soda flake since it is acidic and skin contact irritates.
Why do we need caustic soda flakes?
As we mentioned before, the chemical industry uses caustic soda flake as a raw material. This substance can be used to neutralize acidic environments. As a result of this extraordinary property, it is widely used as a PH regulator. Degreasing is another characteristic of this material. Caustic soda flakes can significantly remove grease. Following are some of its industrial applications:
Food production
Several food processing applications require sodium hydroxide, for instance, water treatment, curing foods like olives, preparing vegetables and fruits for canning by removing their skins.
It is crucial in this case that the method of usage be under scientific principles to be safe for human consumption. Needless to say, this chemical is used in industrial applications and cannot be used domestically.
Pharmaceutical and medicine Industries
The medications and pharmaceutical products containing this substance are pain relievers (aspirin), anticoagulants preventing blood clots, and cholesterol-lowering medicines.
Cosmetic, Sanitary, and cleaning Products
Many detergents and soaps are made from sodium hydroxide, a common ingredient in household and commercial products.
Other industrials
The production of fuel cells relies on sodium hydroxide.
A type of epoxy resin created with sodium hydroxide is used in wind turbines.
Alumina can be derived from naturally occurring minerals by using sodium hydroxide.
video caustic soda flake
SPECIFICATION